Caught up in Christmas spirit
and partying too much yesterday makes me realize I have a young spirit in a
very old body. Today it has let me know I’d overdone it an that’s the reason for
being late in posting this week. With
the season well upon us I’ve chosen to write about some of the science in
picking out a Christmas tree and caring for it.
Of the three kinds of evergreen trees usually used - spruce, fir or pine - I prefer the fir because its needles stay on longer and I find it is the easiest to decorate. If you click on video you’ll
get a few tips on how to choose your tree and care for it. Best to wait until you get the tree home before using a saw to cut off about an inch thick slice from the tree trunk and then put it in a stand that holds water. We don’t think about it much but a
tree is just like a cut flower, to stay fresh it needs to have its freshly cut
end in water.
Any
growing plant needs a constant supply of water to run up the length of its stems to carry nutrients so it can grow and mature. The water evaporates from the
leaves [or needles] and though we are not aware of it, a big leafy tree can give off well
over 100 gallons [~ 400 liters] of water a day. The water climbs up a plant or tree by capillary action, which is just the
ability of a liquid like water to flow up narrow spaces in spite of the force of gravity pulling
on it. We are used to this effect when
we see porous materials like sponges or paper soak up water, or paint being
held between the hairs of a paintbrush.
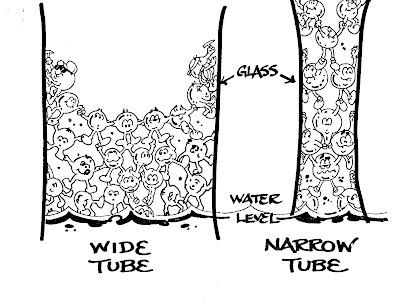
As seen in the diagram, the narrower the tube, the higher the water can
climb. When you consider that the
tallest tree is almost 400 feet [120 meters] in
height - that's figured to be the maximum height possible - you wonder at the power of capillary action!
Being a
chemist by nature, I like to think of the inter-molecular forces that are
responsible for lifting water to such an incredible height. In my
post on water I discussed the uneven distribution of
electrostatic charges around the water molecule that tends to make them stick
together. In the case of plants or trees, the water molecules are attracted to the
sides of pores and also to each other with the result that the combination of these
attractive forces has the power to lift the water to the highest branches of
the tree where the water evaporates causing more water to rise. The fun drawing below shows anthropomorphized water molecules climbing and pulling neighbors with them.
You will
be surprised I think at the amount of water your room temperature Christmas
tree will need to stay fresh. As the water rises and evaporates from the needles it has the added advantage of supplying moisture to the dry air
in your house. Sometimes you are advised to add a nutrient like sugar or aspirin
to the water but the tree is not growing, so it really doesn’t need any. Rie